Introducing the SEBIA HYDRASHIFT 2/4 daratumumab test kit which mitigates daratumumab interferences in multiple myeloma patients treated with Darzalex®.
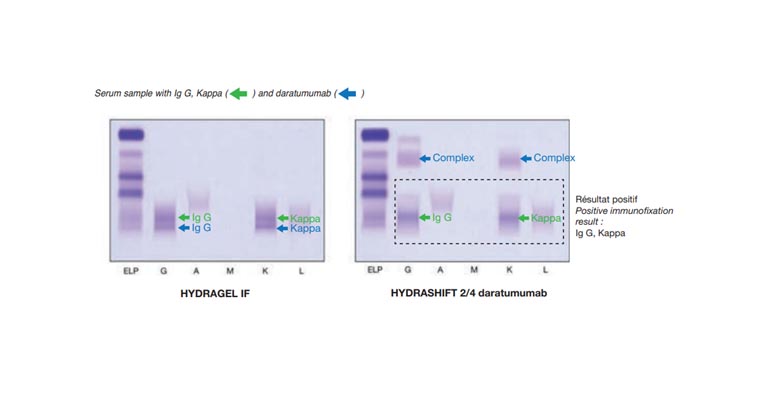
Darzalex ® (Daratumumab) is a fully human anti-CD38 IgG1-k monoclonal antibody used in the treatment of multiple myeloma. The International Myeloma Working Group (IMWG) criteria to assess treatment response is the detection and quantification of monoclonal component (M-Protein) in Serum Protein Electrophoresis and Serum Immunofixation. Case studies show that the Daratumumab antibody simulates an endogenous IgG kappa paraprotein and can be detected as a monoclonal band in a serum immunofixation technique. A significant number of IgG- K M-proteins co migrate with daratumumab which introduces bias in M-Protein quantification and can mask clearance of the M-protein. Therefore, the importance of the Daratumumab peak to not be confused with the M-protein spike is crucial to avoid a false positive result [1].
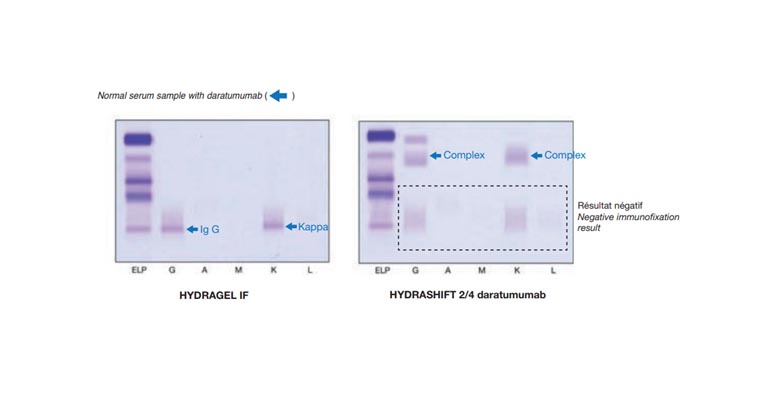
Sebia has developed the HYDRASHIFT 2/4 daratumumab assay in conjunction with their immunofixation assay to detect and separate Darzalex ® interference. The assay procedure uses the addition of anti-daratumumab antigen to bind with daratumumab antibody forming a daratumumab complex. This complex migrates outside the gammaglobulin zone in the alpha-1 zone on IgG and kappa IF tracks. The absence of any monoclonal protein on HYDRASHIFT 2/4 daratumumab immunofixation assay in either Alpha 2, Beta, or Gamma zones is interpreted as a negative assay for monoclonal protein. The presence of a monoclonal (or biclonal) protein on HYDRASHIFT 2/4 daratumumab immunofixation assay in either Alpha 2, Beta, or Gamma zones is interpreted as a positive assay for monoclonal protein
The kit includes the anti-daratumumab antiserum and sample diluent in ready-to-use vials and no sample preparation is required.
References
[1] van de Donk NW, Otten HG, El Haddad O, Axel A, Sasser AK, Croockewit S, Jacobs JF. Interference of daratumumab in monitoring multiple myeloma patients using serum immunofixation electrophoresis can be abrogated using the daratumumab IFE reflex assay (DIRA). Clin Chem Lab Med. 2016 Jun 1;54(6):1105-9. doi: 10.1515/cclm-2015-0888. PMID: 26812873.If you would like to know more about the SEBIA Hydrashift click here or our full range of SEBIA products click here, or alternatively fill in the form below and our Technical Specialists will be happy to help.