
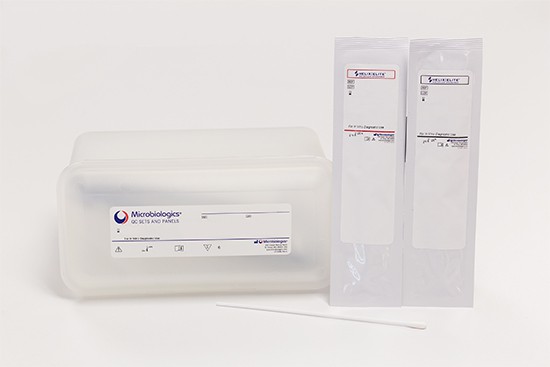
Flu/RSV/SARS-CoV-2 Control Panel (Inactivated Swab)
Details
Flu/RSV/SARS-CoV-2 Control Panel (Inactivated Swab) can be used to monitor the extraction, amplification and detection of Influenza A, Influenza B, Respiratory Syncytial Virus, and SARS-CoV-2 Virus molecular applications. Routine use of external quality controls monitor day to day performance of molecular testing procedures.
Each kit contains six positive and six negative, inactivated control swabs (12 swabs/kit).
Positive Control (six swabs):
Influenza A (H3N2) Virus
Influenza A (H1N1) Virus
Influenza B Virus
Respiratory Syncytial Virus
SARS-CoV-2 Virus
Human lung epithelial cells
Negative Control (six swabs):
Coxsackievirus B1
Human lung epithelial cells
General Information
Biosafety Level 1
Product Format QC Sets and Panels: Helix Elite™
Test Method Molecular Testing
Catalog number 8246
Permits and Restrictions Export Restrictions May Apply
Industry Type Clinical









Reviews
There are no reviews yet.