Why is FLC Measurement so important?
Multiple Myeloma (MM) and other plasma disorders can be characterized by the proliferation of malignant plasma cells. These malignant plasma cells accumulate in the bone marrow and secrete what is known as M-Proteins (Claveau et al., 2022). In normal patients, plasma cells produce excess light chains (Kappa & Lambda) compared to heavy chains.
Not all light chains become incorporated with heavy chains to form immunoglobins, they become known as Free Light Chains (FLC) and can be found in blood serum in small amounts. In the presence of malignant plasma cells, these light chains can be produced in excess levels giving rise to abnormal ratios of serum kappa and lambda FLCs.
The measurement of FLC has become an essential part of the diagnosis and management of plasma disorders such as Multiple Myeloma (MM) (Lutteri, Aldenhoff and Cavalier, 2018). Traditionally the gold standard for the diagnosis of MM has been the use of Serum Protein Electrophoresis (SPEP) and Urine Protein Electrophoresis.
However, limitations based on the half-lives of intact immunoglobulins, mean that SPEP is not sensitive enough to detect small quantities of free light chains (Claveau et al., 2022). In 2001, the serum FLC assay became a routine clinical test for MM as a study found that monoclonal FLCs were found in the majority of Myeloma cases at diagnosis.
The severity of the disease can be determined by the elevation and abnormal ratio of FLC – in turn helping clinicians to monitor disease activity and the patient’s early response to specific therapies. Therefore, proper measurement of free light chains can be an essential part of the early treatment of MM allowing clinicians to tailor therapies specific to their patients (Caillon et al., 2019)
What are the limitations to the current methods?
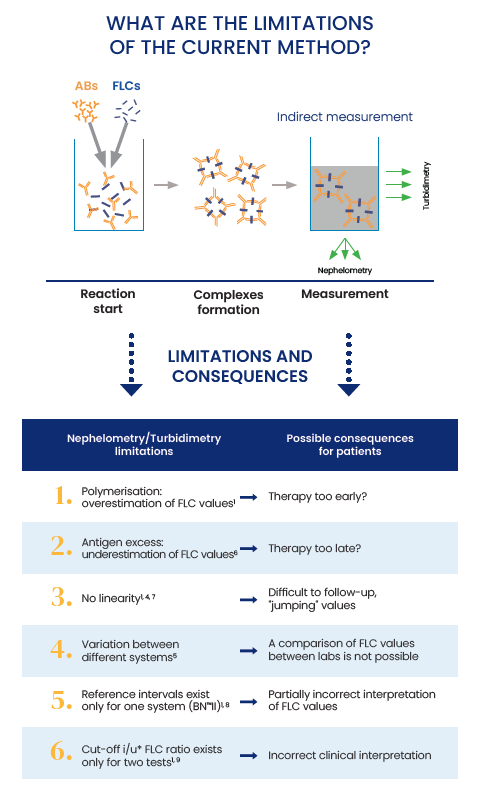
The main assays used for the detection of FLCs are based on nephelometry or turbidimetry technologies. These technologies have their limitations – such as high retest rates, lot to lot variability, limited measuring range, antigen excess giving rise to false low results, non-linearity and overestimations of FLC values (Lutteri, Aldenhoff and Cavalier, 2018).
Recent studies suggest that results gained from the same sample from different assays on different platforms are not equivalent. Due to these analytical limitations, it is essential that the treatment of MM is followed up using the same method throughout the disease on similar platforms (Caillon et al., 2019).
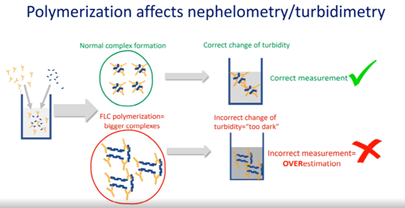
Other methods have issues in regards to Polymerization, which can occur between sFLC leading to the formation of large complexes. These large complexes contain multiple antigenic sites which can cause multiple reactions leading to overestimation of FLC concentration values. This can be observed in both Nephelometry and Turbidimetry methods – with one study showing it can be 10 times higher than the peak estimation (compared to the electrophoretic method).
The same study showed that the Sebia assay for FLCs gave values which were coherent with SPE, demonstrating that the ELISA method is more accurate for the measurement of kappa and lambda FLC (Lutteri, Aldenhoff and Cavalier, 2018).
Antigen Excess is defined as the ≥ 4-fold difference between results obtained with the 2000-fold and 100-fold dilution. Antigen excess can cause the incomplete complex formation of FLC leading to an underestimation of FLC values for assays based on nephelometry and turbidimetry (Vercammen, Broodtaerts, Meirlaen and Bossuyt, 2015).
However, studies suggest that the Sebia FLC assay is not as affected by Antigen Excess compared to other methods as the ELISA method is a direct approach to detecting antibodies with specific antigens. A recent study suggests that Sebia light chain assay uses 3 to 4 times less re-runs compared with other methods, making it both time and cost-effective (Jacobs et al., 2018)
The Advantages to Sebia solution:
As Nephelometric and Turbidimetric have been shown to give results that are not consistent with SPEP/Immunofixation – it is for this reason that Sebia developed the sFLC assay test which ties in perfectly with SPEP/Immunofixation using robust and recognised technology. The Sebia FLC ELISA assay which can be automated on the DAS AP22 Elite platform is an accurate method of testing for serum FLC.
The assay has been shown to have good lot to lot consistency, good precision in terms of reproducibility and the concentrations of FLC were coherent with the peaks observed in SPE (Caillon et al., 2019). This is the major advantage of the Sebia ELISA method as it is the only test that reports FLC values close to values obtained by SPEP when an FLC peak is present. This type of linearity is essential for the ongoing assessment of MM in patients and can be a valuable addition to labs testing for Multiple Myeloma clinics.
Recent studies suggest that the Sebia light chain ELISA assay is not affected by Polymerisation, Antigen excess and has been shown to have good linearity. If set up alongside Sebia SPEP there will be no variation between different systems, which currently don’t allow labs to compare free light chain values between Sebia SPEP & IF with different systems. The coherence between Sebia free light chain and SPE is much stronger compared to other methods (Lutteri, Aldenhoff and Cavalier, 2018) as well as lot to lot consistency (Jacobs et al., 2018). Therefore, Sebia solution for testing FLC is an accurate, time-saving and cost-effective method that would go perfectly hand in hand with customers who already use Sebia SPEP and IF solutions.
Main benefits of the Sebia solution
- Convenient: microplates with breakable strips to fit any lab activity
- Easy-to-use & Fast: simple protocol and only 75min incubation time at RT
- Flexible: validated to use on ELITE (DAS). Can be run on other microplate processors
- Reliable: results concordant with the FLC peak on SPE
- Time and cost-saving: up to 4 times slower retest rate than Freelite on BNII
- Accurate: no underestimation or overestimation on free light chain values versus other methods
Get in touch with our product specialist below for more information on the next generation FLC assay or browse the range here.
Get In Touch
References:
Lutteri, L., Aldenhoff, M. and Cavalier, E., 2018. Evaluation of the new Sebia free light chain assay using the AP22 ELITE instrument. Clinica Chimica Acta, 487, pp.161-167.
Claveau, J., Savary Bélanger, S., Ahmad, I., Delisle, J., De Guire, V., Roy, J. and LeBlanc, R., 2022. Early free light chain reduction following treatment initiation predicts favorable outcome in intact immunoglobulin myeloma. Blood Cancer Journal, 12(1).
Caillon, H., Avet-Loiseau, H., Attal, M., Moreau, P., Decaux, O. and Dejoie, T., 2019. Comparison of Sebia Free Light Chain Assay With Freelite Assay for the Clinical Management of Diagnosis, Response, and Relapse Assessment in Multiple Myeloma. Clinical Lymphoma Myeloma and Leukemia, 19(5), pp.e228-e237.
Vercammen, M., Broodtaerts, L., Meirlaen, P. and Bossuyt, X., 2015. Overestimation of free light chain antigen excess rate. Clinica Chimica Acta, 444, pp.297-302.
Jacobs, J., de Kat Angelino, C., Brouwers, H., Croockewit, S., Joosten, I. and van der Molen, R., 2018. Evaluation of a new free light chain ELISA assay: bringing coherence with electrophoretic methods. Clinical Chemistry and Laboratory Medicine (CCLM), 56(2), pp.312-322.