Cell lines used in the biopharmaceutical industry are often cryopreserved in -80°C freezers short-term or in liquid nitrogen for long-term storage. When the cells are needed for processing, the thawing procedure used can have a huge impact on the recovery, viability and functionality of the cells. Some cryoprotectants, such as DMSO, are toxic above 4°C so it is important to minimise the time cells spend suspended in cryoprotectants at room temperature.
Tips for thawing cells
- Precautions should be taken to prevent injury when collecting and opening vials stored in liquid nitrogen
- When removing cryopreserved cells from storage, proceed directly to thawing or place cells in a dry ice container to minimise exposure to ambient temperatures
- Cells should be thawed rapidly e.g., using a 37°C water bath, to minimise damage to cell membranes
- Use the aseptic technique and work in a biological safety cabinet
- Thaw the vials until only one or two small ice crystals remain (1-2 minutes)
- Wipe cryovials with a tissue soaked in 70% alcohol prior to opening
- Thawed cells should be diluted using pre-warmed culture media
- Determine cell density and viability using trypan blue and cell counter
- Removal of cryoprotectant is dependent on the cell line used. If your cell line is negatively affected by DMSO, wash the cells in pre-warmed media before transferring to a flask
Risks of using water baths
- Thawing is not controlled or reproducible
- Water-based thawing increases risk of contamination
- Water is not suitable for use in cleanroom environments
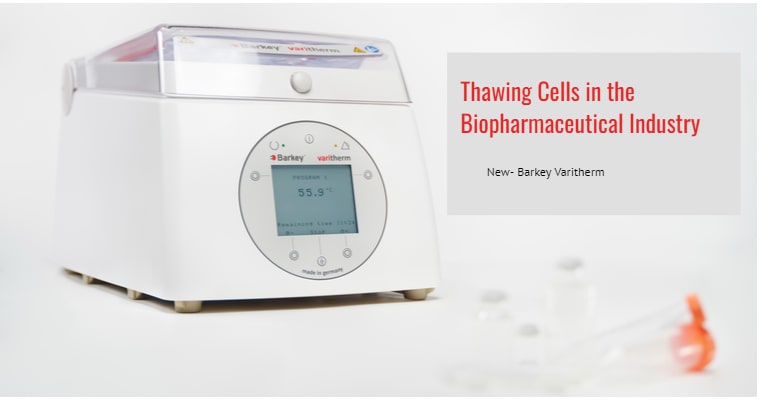
Barkey Varitherm – optimise your cell thawing process
Varitherm water-free thawing system allows you to thaw any container (cryovials, bottles and bags) between cushions filled with water. This dry-heat technology removes the risks associated with water baths and can be used in GMP clean rooms. Water is heated to a pre-defined temperature and circulated through the cushions which conform snugly to the shape of your containers. This results in controlled, water-free thawing which is consistent and reproducible.

Varitherm requires minimal maintenance and the water cushions need to be refilled only once per year using distilled / deionised water and Micropur tablets. The unit is also easy to clean and disinfect, regular laboratory cleaning agents can be used on internal and external surfaces.
Varitherm offers the option of electronic documentation of the warming or thawing process. The Barkey TCP Logging Tool can be used to document barcodes, device users, time and temperature of the executed program.
Using Varitherm gives you the freedom you need:
- Even, consistent temperature and time-controlled thawing
- Easy adjustment of time and temperature settings
- Uses dry heat technology – no contamination
- GMP and Clean Room compliant
- Wide range of heating capabilities between +30°C and +60°C
- Paddles mimic the manual swirling process of water baths
- Provides consistency and reproducibility
- Can also be used for warming culture media or heat inactivation protocols
- Clear lid for visual inspection of your product as it thaws
- Built in ethernet connection
- Leakage detection sensors
- Electronic and mechanical overtemperature alarms
Varitherm is perfect for pre-clinical laboratory use and technology transfer. For biopharmaceutical companies that want a system for both manufacturing and bed-side thawing of cellular products, Barkey also offers the Plasmatherm C&G (Cell & Gene), (download flyer below) which is a medical device that can also be used in hospitals for administration. Plasmatherm C&G is the best choice for customers that intend to write a thawing device into their protocols.
For more information on these products, contact your account manager or get in touch with our dedicated team of product specialists below.
Get In Touch